Corresponding Author: Carlos Alberto Camacho Palacios, General Surgeon of the National University of Colombia. Gastrointestinal Surgeon and Digestive Endoscopist of the Pontifical Javeriana
University. Nutritionist. Postgraduate in Psychology and Psychosomatic Diseases. Former Professor of the Department of Surgery at the National University of Colombia. Director of the MEDINBLUE Center of Gastroenterology, Nutrition, Exercise, and Wellness.
Copy Right: © 2022 Carlos Alberto Camacho Palacios, This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Received Date: November 23, 2022
Published Date: December 01, 2022
Abstract
The gut-brain axis is a bidirectional pathophysiological model that has been studied in the last decades as the origin of multiple diseases. Its dysfunction has been related to the appearance and development of irritable bowel syndrome, dyspepsia, inflammatory bowel disease, obesity, anxiety, depression, fibromyalgia, and even Alzheimer’s disease and cancer. Several studies allow us to take this gut-brain axis to a broader system that also includes the microbiota, nutrients, and metabolism, which we will call the “gut-brain- nutrients-microbiota-metabolism system”. The elements of this system are multi- directionally related, and their dysfunction has in common the alteration of the intestinal barrier, its permeability, immune alteration, and the development of some degree of low- grade inflammation. In this article, we review the five elements of this system, how they are related in their balanced and dysfunctional state and propose a new and integral structure that serves to act on these five objectives by balancing the entire system and thus obtain better results in the prevention and development of related diseases.
Keywords: gut-brain axis, functional gastrointestinal disease system, irritable bowel syndrome, obesity, low-grade inflammation, intestinal barrier disruption, 7-zones program, gut-brain-nutrients-microbiota-metabolism system.
Abbreviations
GBNM2: Gut-Brain-Nutrients-Microbiota-Metabolism
System IBS: Irritable Bowel Syndrome
GBA: Gut-Brain Axis
HPA: Hypothalamus-Pituitary-Adrenal Axis,
MFB: Muscle, Fat, and Bone,
SCFAs: Short-Chain Fatty Acids
IMB: Intestinal Mucosal Barrier
Introduction
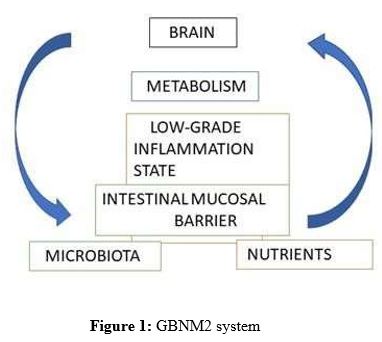
In the last decades, we have gone from being an individual to an organism (holobiont) [1] containing multiple microorganisms (the microbiota) with which we share our genes, nutrients, mediators, and metabolic pathways. How we relate to our bacteria depends on the integrity of the intestinal barrier, the degree of inflammation of the intestinal mucosa, and at the systemic level. We have gone from having a gut-brain axis (GBA) to being a system or better a metasystem (supra system containing other subsystems) with a major information highway between the brain and the gut. [2]
We behave as a gut-brain-nutrients-microbiota-metabolism system (GBNM2), its balance is orchestrated by a proper communication of the whole system, between emotions, the type of microbiota, the intestinal barrier, the nutrients, and the type of metabolism defined not only by diet but also by body composition. Any change in one of them modifies the whole system.
This system behaves in two ways adapted to its dysfunctional state or state of comfort in discomfort (distortion in communication) or in its functional form adapted to its state of well-being.
The dysfunction of the GBNM2 system is related to a common factor which is the inflammation and alteration of the intestinal barrier with increased permeability, activation of the immune system, and with the appearance of functional gastrointestinal diseases, such as irritable bowel syndrome (IBS), dyspepsia, metabolic diseases — as diabetes and obesity —, liver disease, emotional disturbances — as anxiety and depression —, even with fibromyalgia, Alzheimer’s disease, and cancer. [3-8]
There are five elements that interact in this GBNM2 system, as follows.
Brain-emotional system
Brain networks are activated by internal stimuli (interoception, proprioception, cognitive stimuli) and external stimuli (through the senses) generating emotional behavioral strategies that can be defensive
— hypothalamus-pituitary-adrenal (HPA) axis — or emotional strategies of growth, well-being, and integration through conscious networks.[9] There are brain networks that participate in the internal dialogue (default network), when we are interested in a goal (executive network) or when we seek a reward (reward network). The brain connects and disconnects from the different networks according to the area or strategy we are interpreting, that is why different connectivity in depression, in stress, or in falling in love. [10, 11, 12]
Since childhood, the brain has built strategies that become algorithms with which it will automatically respond to different daily events. These predesigned emotional strategies allow the brain to act quickly, and unconsciously, saving energy. These algorithms with which our brain compares reality can be correct or incorrect. The communication of the GBNM2 system is done in several ways:
Bidirectional neuronal pathway – through the communication of the brain with the autonomic nervous system via the vagus nerve. [13]
Immune pathway – through the intestinal immune system. [14]
Endocrine pathway – by the cerebral neuroendocrine system (HPA) and by the production of metabolites of the intestinal microbiota that behave like hormones which circulate in the body and reach other organs. [15, 16]
Metabolites production – such as SCFA (butyric and propionic acid) by bacteria that can act at the brain level. [17, 18, 19, 20]
Intestine
Participating in this communication network or GBNM2 system are the intestinal barrier, the enteric nervous system (myenteric and submucosal plexus), the epithelial cells of the intestinal mucosa with their intercellular junctions, immune cells, dendritic cells, macrophages, lymphocytes, and plasma cells. [21]
Microbiota
The gut microbiota is a complex community of 100 trillion microbes that are more than 10 times the number of cells in the body and have more than 150 times more genes than those in our genome. [22]
The microbiota produces many neurotransmitters such as catecholamines, GABA, and tryptophan that impact the hypothalamus. [23, 24]
The microbiota is considered as if it were another organ of the individual or an ecosystem capable of influencing brain function and regulation, metabolism, and the degree of inflammation. It can change neuroendocrine function [25] and can affect brain physiology, behavior, and cognitive function. [20, 26, 27, 28] It also has an important influence on the brain through the neural network, the neuroendocrine system, and the immune system [29] and regulates energy expenditure and appetite. [30]
The microbiota produces short-chain fatty acids (SCFAs) that regulate intestinal functions including (free fatty receptor) motility, secretion, and inflammation. [31] It stimulates enteroendocrine production of serotonin 5-HT, [32] which acts in the enteric nervous system modulating motility, secretion, and visceral hypersensitivity, and at the central nervous system (CNS) level it acts modulating mood. Serotonin modulates symptoms related to IBS. [33, 34]
Nutrients
The type of nutrients we choose from the diet will define the composition of the microbiota and its metabolism [34], body composition, and energy expenditure. Alcohol consumption alters the composition and function of the microbiota and the host intestinal barrier. [35- 39] The diet can also alter intestinal permeability. [40]
Metabolism
A proper body composition and hormonal balance are also part of the GBNM2 system. Exercise makes the cells work better, improves the quality of mitochondria and cellular respiration, [41, 42] and has an anti-inflammatory effect. [43] When a proper ratio between muscle, fat, and bone content (MFB balance) is achieved, there is better management of caloric expenditure.
Dysfunctions of the GBNNM2 system emotional dysfunction
Our brain is also involved in metabolic balance through the neuroendocrine axis and is in permanent communication with the microbiota and its genetic load.2 The amygdala is located deep in the temporal lobe of the brain and regulates defensive emotions. [44, 45, 46] Stress induces alterations in the neuroendocrine and immune pathways of the GBA.[47] This causes changes in the sensitivity of peripheral immune cells resulting in an imbalance between pro- and anti-inflammatory responses [48] and induces dysbiosis. [49]
Stress can cause hyper- or hypoactivity of the HPA axis, the autonomic nervous system, and the metabolic and immune systems, altering the GBA and leading to alteration of the physiological functions of the gastrointestinal tract. [50] It affects the composition and growth of the microbiota [50], and visceral [52] Acute and chronic stress alters the microbiota, intestinal mucosal immune cells, and neurotransmitter production.[47] Stress can lead to disruption of the sympathetic and parasympathetic nervous system of the HPA axis, endogenous pain modulation, and the ascending aminergic pathway. [53] It can also develop or aggravate IBS symptoms.[54]
Excessive activation of the amygdala has been reported to cause mood disorders and cognitive dysfunction. [55, 56] There is evidence of autonomic dysfunction and impaired HPA axis activity in patients with IBS and inflammatory bowel disease (IBD). [58] Stress induces alterations in the microbiota accompanied by changes in cytokines and chemokines. [57, 59] Stressful moments also alter the microbiota through glucocorticoid production. [60, 61, 62]
Stress and the HPA axis affect the composition of the microbiota. Stress in childhood can lead to changes in the microbiota and this becomes a risk factor for stress-related disorders in adult life. [63, 64] Early stress and maternal separation could lead to long-term changes in the HPA and microbiota. [65, 66] Rodent studies showed that stress changed the barrier function of the intestinal mucosa, caused LPS and other cytokines to enter the blood circulation, and stimulated TLR4 and other TLRs that produce inflammatory cytokines. Peripherally produced inflammatory factors could increase the permeability of the blood-brain barrier, making it possible for peripherally produced inflammatory factors to directly influence the brain.[67]
The brain acts through the autonomic nervous system and the HPA axis in the digestive system including transit, motility, on the intestinal mucosal barrier (IMB) for secretion and permeability, and on the expression of the microbiota. [68]
Emotional disturbances predispose to alterations in bowel function and in turn, bowel disruption predispose to emotional disturbances, as seen in studies of IBS. [69, 70, 71] Stress induces intestinal inflammation, in animal models and humans. Stress and intestinal infections can alter the IMB and encourage inflammation, [72, 73] as well as leading to IMB dysfunction with motor abnormalities and visceral pain as seen in IBS.74, 75 Stress can increase intestinal permeability [51, 76, 77, 78] by mast cell mechanisms and by elevation of cortisol, [51, 79, 80] which can lead to bacterial translocation and mucosal inflammation [73] and induce activation of intestinal mast cells.[81, 82, 83] There is a vicious circle between chronic stress, activation of peripheral stress systems, psychiatric comorbidity, and gastrointestinal symptoms.[52]
Intestinal dysfunction
The emotional system has a great impact on the intestine (autonomic nervous system — ANS), on its microbiota [84], and on the permeability of IMB, likewise changes in the microbiota and alterations in IMB cause alterations and activation of the immune system with a certain degree of low-grade inflammation. [85, 86] The central nervous system and the enteric system can also modulate the microbiota through changes mainly in the function of the intestinal wall. [2, 87]
Nutritional dysfunction
Changes in diet can alter the microbiota and this, in turn, alters the intestinal mucosal layer allowing access to microbial particles that come in contact with dendritic cells increasing the permeability of the IMB — releasing mediators that lead to immune activation in different organs including the brain (metabolic endotoxemia) [88] — resulting in low-grade inflammation that has been linked to several diseases.
Diets with fast and processed foods have been associated with increased intestinal permeability and depressive symptoms. On the other hand, diets rich in vegetables, fruits, and fish are associated with lower depressive symptoms. [89]
Diets high in fat cause the expression of more gram-negative bacteria with high levels of LPS [90] (metabolic endotoxemia) which is related to inflammation of the intestine, liver, adipose tissue, and in diabetes [91] and are capable of altering the microbiota. [92]
Microbiota dysfunction
The microbiota is an independent organ [95] and is also a complex system of organisms [96] connected to all other organs in the system by bidirectional neural, immune and endocrine pathways68 that will influence CNS and enteric development, [97] intestinal motility, [98] immune system, nutrition, degree of intestinal inflammation, and can also cross the IMB when it becomes permeable and affect other organs at a distance. The microbiota modulates various host physiological functions including glucose metabolism and liver function [99] and acts on brain development.[100] It may also affect the Enteric Nervous System [101] and participate in mood and behavioral changes, such as depression, through microbial metabolites (SCFAs) and generate proinflammation (LPS) [102, 103] through the vagus nerve104 and through the production of neurotransmitters such as serotonin (5-HT).[105] The microbiota affects neurogenesis and hippocampal gene expression, thus participating in neuroplasticity.[106] GABA-producing bacteria have been shown to alleviate depression and anxiety behavior.[107]
The particular genetics of an individual is also a factor that selects the type of microbiota [108] and different cultures, since different diets present a different type of microbiota.[57, 109, 110]
The microbiota is capable of modifying behavior including sexual and social behavior, stress, learning, memory, and eating and obesity behaviors.[111]
Depression has been associated with the alteration of the microbiota, and the transfer of microbiota from depressive rodents to healthy rodents induces depressive behaviors in the recipients suggesting the role of the microbiota in the pathophysiology of depression. [112, 113]
Alteration of the microbiota is related to chronic diseases such as asthma, IBD, depression, obesity, type 2 diabetes (insulin resistance, inflammation),[114, 115] cardiometabolic complications, [8] and even cancer.[116]
Metabolism dysfunction
The fifth element of the GBNM2 system is the type of metabolism, which is the process of the organism to obtain or produce energy. It depends on the food in the diet, the state of the hormonal system, and the body composition, i.e. the MFB balance and the physical activity that determine the metabolic rate.
Obesity can decrease the quality of life and increase the frequency of mental disorders, such as depression and anxiety.[117] The network of neurons and hormones between the brain and the gastrointestinal tract act on receptors that regulate appetite, food intake, and obesity.[118] The microbiota of an obese person provides more energy to the host than the microbiota associated with leanness.[119]
The GBNM2 system adapts to the internal and external environment in a dysfunctional and functional manner.
Description of how this dysfunctional GBNM2 system behaves:
When there is an imbalance in one or more of its components, the whole system suffers and manifestations of pain and hypersensitivity appear in various organs. Symptoms of IBS and dyspepsia overlap with other non-gastrointestinal conditions such as fibromyalgia, chronic fatigue syndrome, overactive bladder, chronic pelvic pain, and other chronic pain syndromes. They all fit into the same bio-psycho-social pathophysiological model.[120]
Due to the multifactorial pathogenesis of functional gastrointestinal disease, psychological and lifestyle intervention, self-help strategies, and nutritional approaches are required in addition to pharmacological therapy.[121]
Dysfunction of the GBNM2 system is accompanied by dysfunction of its subsystems, as follows.
- Emotional dysfunction: an emotional blockage with the alteration of the stress axis. HPA with hyperactivation or hypoactivation of the sympathetic system. It then appears a greater tendency to mood disorders with depression, anxiety, and exhaustion, since that stress alters the microbiota and intestinal permeability. When the brain uses its predictive system erroneously.
- Intestinal dysfunction: disruption of the intestinal barrier. Increase of intestinal permeability, altering the intestinal cells and their neurotransmitters. Appearing an environment conducive to bacterial translocation. Alteration of the enteric nervous system with visceral hypersensitivity.
- Microbiota dysfunction: alteration of the composition and growth of the microbiota, knowing that chronic stress induces dysbiosis.
- Immune dysfunction: all the previous leads to a low-grade inflammation with immune activation of the intestinal wall, with the release of mast cells, mediators, and cytokines. When stress is prolonged there will be an increase in cortisol and chronic systemic inflammation. Dysfunction of the intestinal barrier and of the microbiota leads to a certain degree of inflammation which has repercussions on the brain and the blood-brain barrier (neuroinflammation).
- Nutritional dysfunction: contributing to this is a diet of fast food and abundant fats that increase intestinal permeability, causing depressive symptoms and altering the microbiota.
- Metabolic dysfunction: as a result, there are changes in body composition with imbalanced MGB with an increased percentage of fat, increased inflammation, endothelial dysfunction and metabolic disorders, such as obesity and diabetes.
- GBNM2 dysfunction: then there is a dysfunction of the entire GBNM2 system, which is the combination of emotional, intestinal, nutritional, microbiota and metabolic dysfunction with the appearance of different manifestations such as IBS, dyspepsia, migraine, anxiety, depression, fibromyalgia, pelvic pain, chronic fatigue, accelerated aging that when related to chronic inflammation may predispose to the development of Alzheimer’s disease and cancer.
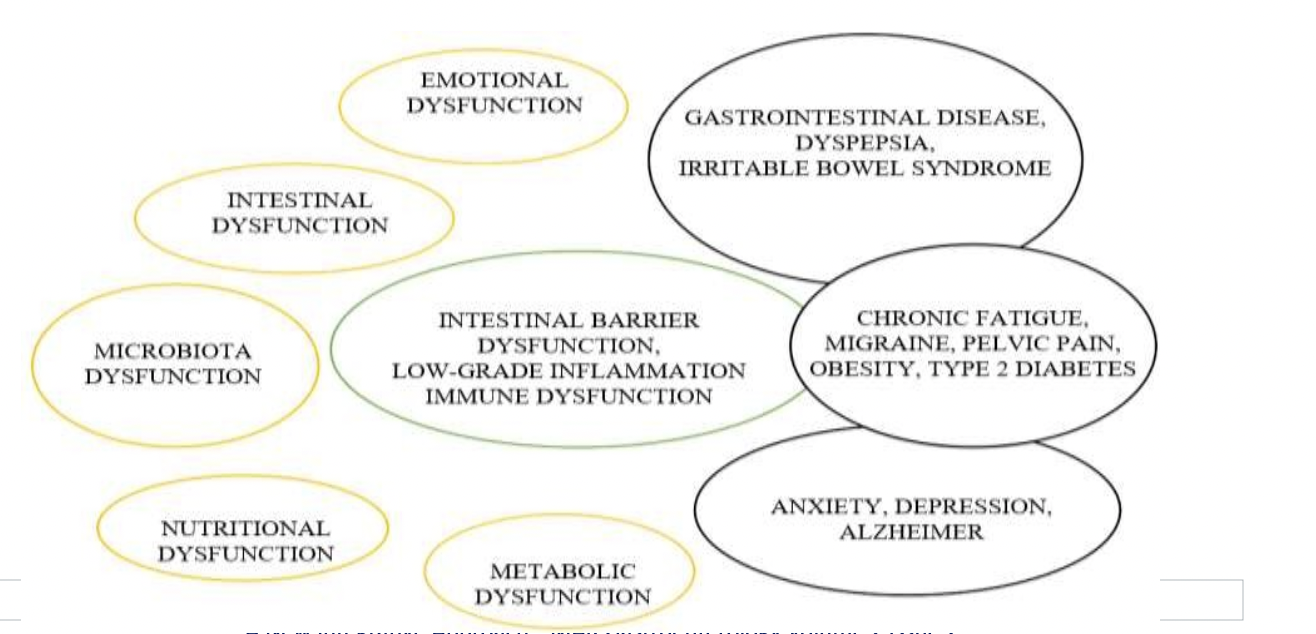
Figure 2: GBNM2 system dysfunction
-
Dysfunction | Alteration | Results |
Emotional dysfunction | – Blockage of the emotional system, alteration of the brain predictive processing model, hyperactivation and hypersensitivity of the central and peripheral nervous system, HPA axis, and vagus nerve – Increased cortisol causes alteration of microbiota and intestinal barrier permeability | – Anxiety, stress, depression, aggressiveness, appetite dysregulation – Alteration of the microbiota, of the intestinal barrier, causing mucosal inflammation, IBS |
Intestinal and Immune Dysfunction | – Alteration of the intestinal barrier with increased permeability and immune system (barrier dysfunction), dysbiosis, cytokine production, intestinal inflammation affecting the mood | – Bacterial translocation – Activation of the intestinal immune system – Low-grade inflammation – Alteration of intestinal motility, hypersensitivity (IBS) |
Microbiota dysfunction | – Alteration in the composition of the microbiota, dysbiosis – Proinflammatory factors through an immune pathway, causing neuroinflammation and cognitive impairment – Decrease of bacterial neurotransmitters and alteration of metabolite production, the microbiota is selected in each disease, e.g. in obesity | – The microbiota imbalance orchestrates the development of neuropsychiatric (neuroinflammation), metabolic and functional disorders – Increased degree of inflammation – Increased efficiency of food calorie intake (obesity)122, endothelial dysfunction, hypertension, heart disease |
Nutritional dysfunction | – Unbalanced intake, high consumption of fat, refined carbohydrates, sweeteners, processed foods, etc. | – Excess fat, abdominal obesity, inflammation |
Microbiota dysfunction | – Alteration in the composition of the microbiota, dysbiosis – Proinflammatory factors through an | – The microbiota imbalance orchestrates the development of neuropsychiatric |
| | immune pathway, causing neuroinflammation and cognitive impairment – Decrease of bacterial neurotransmitters and alteration of metabolite production, the microbiota is selected in each disease, e.g. in obesity | (neuroinflammation), metabolic and functional disorders – Increased degree of inflammation – Increased efficiency of food calorie intake (obesity)122, endothelial dysfunction, hypertension, heart disease |
Metabolic dysfunction | – Excess fat, reduced muscle, and bone tissue, MFB imbalance, increased fat percentage | – Alteration of body composition, abdominal obesity, metabolic syndrome, overweight, inflammation |
Table 1: Dysfunction of the GBNM2 system
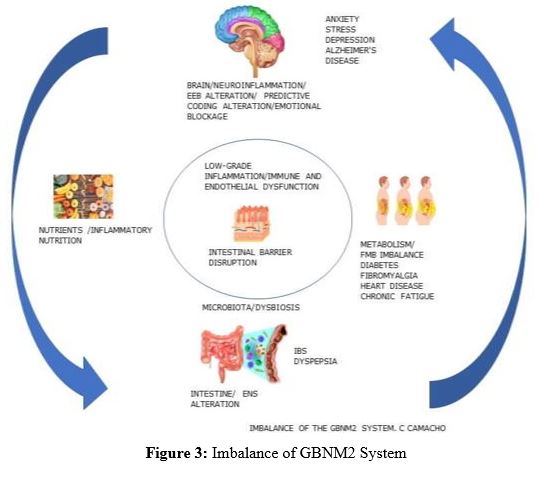 Figure 3: Imbalance of GBNM2 System
Figure 3: Imbalance of GBNM2 System
Therapeutic objectives in the dysfunction of the GBNM2 system
There are five therapeutic objectives to correct the dysfunction of the GBNM2 system, as presented in the following table.
GBNM2 system dysfunction | Therapeutic objectives |
Emotional dysfunction | – Knowing the structure of the emotional system, emotional zones, and strategies, to flow and integrate the whole system, relearning behavioral programs, and intestinal sensitivity. – Psychotherapy can reconfigure predetermined activation patterns89 |
Intestinal and Immune dysfunction | – Natural nutrition, and microbiota balance, restore the intestinal barrier – Control of the degree of inflammation – Prebiotics, probiotics, fermented foods |
Microbiota dysfunction | – Natural and varied nutrition, fermented foods, promoting a diverse microbiota, appropriate use of antibiotics, specific prebiotics and probiotics, symbiotics, fecal matter transplantation |
Nutritional dysfunction | – Natural nutrition, fermented foods, avoiding toxins, processed foods, excess fat, refined carbohydrates, alcohol, and sweeteners |
Metabolic dysfunction | – Balance of body composition (MFB), balance of diet and physical activity |
Table 2: Therapeutic objectives in the dysfunction of the GBNM2 system
A functional emotional system
It is very important to know the emotional system to transform it and enter into a state of growth, self- realization, and well-being. For this, I suggest the 7-zones program, which is a structure where we can intervene in the different levels of the GBNM2 model based on emotional intelligence.[9]
The first zone is our essence, our true nature, which has to do with our fundamental values.
The second zone is the survival zone, it is constituted by all the information that allows us to survive and contains all the defensive strategies to face what we consider a threat, either real or imaginary, and the way we react to them. Being in this zone for a long time causes stress and inflammation in different parts of the body — dyspepsia, IBS, fibromyalgia, and many more —, it is where psychosomatic diseases are expressed. We are usually unaware of being trapped in this area, identified with our defensive selves. It is where defensive emotions, such as aggressiveness, fear, or sadness (a state of comfort in discomfort), manifest themselves.
This zone has two levels — neuro-programming level corresponds to all the information and strategies with defensive emotions that have been created since childhood and neuro-reaction level is the way the individual reacts to a threat with preconceived strategies.
The third zone is the zone of neuro-transformation and resistance to change. It is the zone where we can appreciate, be conscious and take distance from the survival zone and the strategies that cause discomfort, its predictive model and its content (limiting beliefs, filters, meanings, expectations, interpretations, symptoms, needs, identities). The different distortions of language are evaluated and the individual can become aware of his world and his environment; he can modify the models he uses to interpret it, using the principles of cognitive behavioral therapy [123], defuse thoughts from emotions; stop seeing himself not as his defensive self but as the context (the observing consciousness) where reality happens; disidentify himself from the content, accepting pain as part of life, and commit to his values to follow what he most wants from life [124] being aware of his reality in the present (mindfulness).[125]
In this zone of transformation, it is normal that there is resistance to change because the brain does not like to change the networks to which it has been adapted for a long time and from which it obtains a reward, it tries to protect the person from being rejected, from making a mistake or from anxiety.126 Change is a process that goes from not knowing that you need to change, to knowing that you need to change, then doing it, and then staying in that new state.[127]
The brain is probabilistic, it creates an adaptive model of the internal and external world, its best hypothesis. If we learn new strategies the brain will make different simulations – it will take new algorithms as predictive models towards well-being – the brain continuously predicts what is going to happen using already pre-designed strategies (internal models) to maintain allostasis (adaptation to environmental demands and energy balance) if these strategies are not correct (altered predictive models) its actions will cause discomfort and pain (as in IBS where there may be a continuous perception of pain even though the sensory sensations of the gut are normal or in depression where
there may be interrupted reward prediction errors) with an incongruence between brain perception and interoceptive bottom-up information, then it should re-evaluate its predictive model (updating the model and its emotional strategies) to correct this predictive error (learning). [128-132]
Some consider that the gut is capable of learning. Stress could alter perception through a process of learning and memory. Opening a door to cognitive-behavioral therapies to extinguish learning as in the case of IBS.[120]
Zone four is the zone of growth, development, and well-being, it is easier to enter it if we have already overcome zone three. It has two levels — neuro-design and neuro-wellness. The level of neuro-design is the level of self-realization where a journey is made from the values (what is most important to the person) to the goals that have been proposed. Letting oneself be carried by a creative tension that results from starting from the current state to reach the desired state.[133] The second level of this zone is where the person can use their strengths, virtues, and positive emotions such as gratitude, joy, compassion, optimism, and empathy to enter a state of plenitude, taking advantage of the positive aspects of the human experience, avoiding concentrating only on repairing the negative and traumatic aspects of existence.[134]
The fifth zone is the neuro-connection zone, when developing this zone the values are aligned with the goals and with a life purpose which is the legacy you want to leave to others, something great, good, and positive for the world. Humans are characterized by the search for meaning, for self- transcendence, for a life purpose through consciousness.[135]
Zone six, the zone of neuro-integration is where all the zones become conscious, where you can move with flexibility in all of them and not remain blocked in the zone of survival. From here you can contemplate the whole mental model, the defensive, creative, and wellbeing strategies that you are using and decide if you want to make changes that allow you to have a better life experience.
Zone seven is the body zone, it is where the emotions are reflected in the organs of the body, if there is discomfort there will be symptoms, if it enters the wellness zones and flows throughout the system the symptoms will have no value. The result is the balance of the GBNM2 system in a state of comfort in wellness.
Zone | | Intervention | Impulse |
Zone 7 | Body | | - Homeostasis, control of inflammation, regulation ofmetabolism
|
Zone 6 | Neuro-integration | - Observation, awareness, and fluidity in all emotional areas
| - Vision of the system
- Protective, holistic,
nurturing, empowered self
|
Zone 5 | Neuro-connection | - Alignment of values, goals,
and purpose
| |
Zone 4 (growth zone) | Neuro-wellness level | - Focus on strengths, virtues, and positive emotions
- Mindfulness
| |
Zone 4 (growth zone) | Neuro-design level | - Creative strategies
- Moving from the current state to the desired state
| |
Zone 3 | Neuro- transformation zone and resistance to change | - Becoming aware of and distancing oneself from defensive strategies
- Process of change
- Correction of wrong predictive strategies
| - Observing and transforming self, emotional liberation
|
Zone 2 (survival zone) | Neuro-reaction level | - Acting in a reactive, defensive manner
- Unconscious patterns
| - Defensive self, contained self, dramatic, historical, critical self
|
Zone 2
(survival.
zone) | Neuro- programming level | - Psychobiography
- Creation of defensive strategies/ predictive strategies
| |
Zone 1 | Essence | | |
Table 3: 7-zones program
Adapted from the 7-zones program from the book “La estrategia del bienestar” (The Wellbeing Strategy), by Carlos Alberto Camacho Palacios.[9]
When you transcend all the zones you enter a state where you can flow in wellness, in complete homeostasis of the whole system.
A Functional Gastrointestinal System
In this system, we will focus on all the actions we can do to maintain and restore the intestinal barrier, to control and balance its state of inflammation and prevent bacterial translocation and immune disruption.
A Designed Nutrition
Nutrition should be based on natural foods, with plenty of fiber, fruits, vegetables, fermented foods, and water, avoiding excess of saturated fats mainly, excess of refined carbohydrates, additives, and sweeteners. Supplements are necessary when there is a deficiency or when we want to support a specific function of the organism. Designed nutrition means that the diet must be personalized as all foods may not be suitable for all individuals considering the underlying pathologies.
Restoring the Microbiota
It is important to keep a stable and diverse gut microbiota as it helps maintain the immune system, brain development, intestinal barrier, metabolism, appetite control, mood, and disease prevention by reducing the magnitude of inflammation.[136] The type of diet will depend on the type of microbiota.[137] If we consume protein and fat, this will increase the bacteroids; on the other hand, if
we consume carbohydrates, it will increase the genus prevotella (MetaHIT).[138] Dysbiosis has been associated with the development of anxiety, depression, neuroinflammation, autoimmune disease,
[139] obesity, diabetes, and heart disease [140] and influences the energy balance of the host. [141] The microbiota of non-obese individuals has shown to be more diverse than that of obese individuals. [142]
Prebiotics promote the growth of beneficial bacteria in the gut. [143] Also, prebiotics have an anti- obesity impact by reducing the proinflammatory state, elevating SCFAs production, and improving the intestinal barrier. [22, 144]
On the other side, probiotics may even correct the harmful effects of microbiota alteration and inflammation.[145] They help to restore the intestinal barrier146 and tight junctions between epithelial cells, reducing permeability, preventing bacterial translocation, and reducing lipopolysaccharide- derived inflammation.[147, 148] As an example, bifidobacteria have demonstrated their ability to improve intestinal barrier function in several studies.[149] Probiotics may have effects in regulating overweight, but this appears to be strain and species-specific.[150] They are a tool that can help decrease the problem of obesity.[8, 151]
Symbiotics modulate metabolic activity in the gut [152] and help in the control of blood sugar and fat levels and in the prevention of osteoporosis.[153]
Fecal transplantation may be useful in the treatment of chronic constipation, irritable bowel syndrome, Crohn’s disease, ulcerative colitis, obesity, and type 2 diabetes.[154]
The composition of the microbiota may be helpful in the comprehensive treatment of the balance of the GBNM2 system, but more research is needed to verify the efficacy of this therapeutic approach.
The metabolic factor
Diets high in sugar and fat cause damage to cellular metabolism (mitochondrial dysfunction) while exercise makes the cell work better, and improves the quality of mitochondria and cellular respiration [42, 155]
The MFB balance consists of bringing the body through aerobic/anaerobic type exercise at least three times a week to a composition with an adequate amount of muscle, a correct percentage of fat, and maintaining a good bone density, thus facilitating an active metabolism and a controlled inflammatory state.
Conclusion
The connection between the brain and the intestine called the “gut-brain axis” can be expanded to a gut-brain-nutrients-microbiota-metabolism system; it goes from being a bidirectional axis to a multidirectional system, where all these subsystems interact and any dysfunction in any of them alters the whole superorganism that is the individual. The prevention and treatment of diseases should seek the recovery and balance of all five areas of the GBNM2 system simultaneously.
To intervene in the emotional system, it is necessary to do it from a structure that allows us to address all the emotional areas of the individual and their impact on the body. For this, this article proposes the 7-zones program where the emotional balance is integrated considering the different psychological theories of greater effectiveness as cognitive behavioral therapy, acceptance and commitment therapy and positive psychology, in addition to the correction of the predictive brain model, as well as the recovery of the intestinal barrier to regulate the state of inflammation using a natural and balanced diet, prebiotics and selected strains of probiotics, and thus obtain a diverse and appropriate microbiota for each individual. Regarding the search for a balance in body composition in terms of the percentage of muscle, fat, and bone tissue (MFB balance), continuous aerobic and anaerobic exercise is suggested. All this is to correct the dysfunctionality of the GBNM2 system and bring the individual to his best state of well-being. Restoring the balance of the GBNM2 system (7-zone program) by optimizing each of its subsystems is a comprehensive way to prevent and treat diseases such as dyspepsia, irritable bowel syndrome, obesity, fibromyalgia, anxiety, depression, Alzheimer’s, and cancer. We know that all of them have a multifactorial origin and with common factors in their pathophysiology (inflammation and alteration of BMI), therefore, they require a comprehensive management. Although evidence is growing on how these subsystems are related, there is still a lack of clarity on their underlying mechanisms and how we can influence them for the benefit of health. Careful clinical trials must be designed to have more solid evidence.
Reference
- Margulis L.Fester, R(Eds.) Symbiosis as a sourse of evolutionary innovation:Speciation and Morphogenesis. Cambridge, MIT Press;1991.
- Osadchiy V, Martin CR, Mayer EA. Gut microbiome and modulation of CNS function. Compr Physiol. 2019 Dec 18;10(1):57-72.
- MacFarlane GT, Macfarlane LE. Acquisition, evolution and maintenance of the normal gut microbiota. Dig Dis. 2009;27 Suppl 1:90–8.
- Scanlan PD, Shanahan F, Clune Y, Collins JK, O’Sullivan GC, O’Riordan M, et al. Culture- independent analysis of the gut microbiota in colorectal cancer and polyposis. Environ Microbiol. 2008 Mar;10(3):789–98.
- Brun P, Castagliuolo I, Di Leo V, Buda A, Pinzani M, Palù G, et al. Increased intestinal permeability in obese mice: new evidence in the pathogenesis of nonalcoholic steatohepatitis. Amer J Physiol Gastrointestinal Liver Physiol. 2007 Feb; 292(2):G518–25.
- Creely SJ, McTernan PG, Kusminski CM, Fisher FM, Da Silva NF, Khanolkar M, et al. Lipopolysaccharide activates an innate immune system response in human adipose tissue in obesity and type 2 diabetes. Am J Physiol Endocrinol Metab. 2007 Mar;292(3):E740–7.
- Ley RE, Bäckhed F, Turnbaugh P, Lozupone CA, Knight RD, Gordon JI. Obesity alters gut microbial ecology. Proc Natl Acad Sci USA. 2005 Aug 2;102(31):11070–5.
- Shen J, Obin MS, Zhao L. The gut microbiota, obesity and insulin resistance. [Internet]. Vol. 34, Molecular Aspects of Medicine. 2013. p.39–58. Available from: http://dx.doi.org/10.1016/j.mam.2012.11.001
- Camacho CA. La estrategia del bienestar. Penguin Random House-Caligrama; 2020:16-32
- Van Oort J, Tendolkar I, Hermans EJ, Mulders PC, Beckmann CF, Schene AH, et al. How the brain connects in response to acute stress: a review at human brain system level. [Internet]. Vol. 83,Neuroscience & Biobehavioral Reviews. 2017. P.281-297. Available from: http://dx.doi.org/10.1016/j.neubiorev.2017.10.015
- Shi L, Sun J, Xia Y, Ren Z, Chen Q, Wei D, et al. Large-scale brain network connectivity underlying creativity in resting-state and task fMRI: Cooperation between default network and frontal- parietal network. Biol Psychol. 2018 May;135:102-111.
- Li BJ, Friston K, Mody M, Wang HN, Lu HB, Hu DW. A brain network model for depression: From symptom understanding to disease intervention. CNS Neurosci Ther. 2018 Nov;24(11):1004- 1019.
- Bravo JA, Forsythe P, Chew MV, Escaravage E, Savignac HM, Dinan TG, et al. Ingestion of Lactobacillus strain regulates emotional behavior and central GABA receptor expression in a mouse via the vagus nerve. [Internet]. Vol. 108, Proceedings of the National Academy of Sciences. 2011. p. 16050‐16055. Available from: http://dx.doi.org/10.1073/pnas.1102999108
- Agustí A, García‐Pardo MP, López‐Almela I, Campillo I, Maes M, Romaní-Pérez M, et al. Interplay between the gut‐brain axis, obesity and cognitive function. Front Neurosci. 2018 Mar 16;12:155.
- Cussotto S, Sandhu KV, Dinan TG, Cryan JF. The Neuroendocrinology of the Microbiota‐Gut‐ Brain Axis: A Behavioural Perspective. Front Neuroendocrinol. 2018 Oct;51:80‐101.
- Schroeder BO, Bäckhed F. Signals from the gut microbiota to distant organs in physiology and disease. Nat Med. 2016 Oct;22(10):1079‐1089.
- Erny D, Hrabě de Angelis AL, Jaitin D, Wieghofer P, Staszewski O, David E, et al. Host microbiota constantly control maturation and function of microglia in the CNS. Nat Neurosci. 2015 Jul;18(7):965‐ 977.
- De Vadder F, Kovatcheva‐Datchary P, Goncalves D, Vinera J, Zitoun C, Duchampt A, et al. Microbiota‐ generated metabolites promote metabolic benefits via gut‐brain neural circuits. Cell. 2014 Jan 16;156(1-2):84‐96.
- Schmidt C. Mental health: Thinking from the gut. Nature. 2015 Fev 26;518(7540):S12– 5.
- Mayer EA, Knight R, Mazmanian SK, Cryan JF, Tillisch K. Gut microbes and the brain: Paradigm shift in neuroscience. J Neurosci. 2014 Nov 12;34(46):15490–6.
- Santis SD, De Santis S, Cavalcanti E, Mastronardi M, Jirillo E, Chieppa M. Nutritional Keys for intestinal Barrier Modulation [Internet]. Vol. 6, Frontiers in Immunology. 2015. Available from: http://dx.doi.org/10.3389/fimmu.2015.00612
- Chen J, He X, Huan J. Diet Effects in Gut Microbiome and Obesity [Internet]. Vol. 79, Journal of Food Science. 2014. p. R442-451. Available from: http://dx.doi.org/10.1111/1750- 3841.12397
- Russell WR, Hoyles L, Flint HJ, Dumas ME. Colonic bacterial metabolites and human health [Internet]. Vol. 16, Current Opinion in Microbiology. 2013. p. 246-254. Available from: http://dx.doi.org/10.1016/j.mib.2013.07.002
- Lyte M. Microbial endocrinology: Host-microbiota neuroendocrine interactions influencing brain and behavior. Gut Microbes. 2014 May;5(3):381–9.
- Farzi A, Frohlich E,Holzer P. Gut Microbiota and the Neuroendocrine System.Neurotherapetics 2018;15:5-22
- Diaz Heijtz R, Wang S, Anuar F, Qian Y, Björkholm B, Samuelsson A, et al. Normal gut microbiota modulates brain development and behavior. Proc Natl Acad Sci USA. 2011 Feb 15;108(7):3047–52.
- Jenkins TA, Nguyen JCD, Polglaze KE, Bertrand PP. Influence of tryptophan and serotonin on mood and cognition with a possible role of the gut-brain axis. Nutrients [Internet]. 2016 Jan 20;8(1). Available from: http://dx.doi.org/10.3390/nu8010056
- Cryan JF, Dinan TG. Gut microbiota: microbiota and neuroimmune signalling Metchnikoff to microglia. Nat Rev Gastroenterol Hepatol. 2015 Sep;12(9):494–496.
- Ogbonnaya ES, Clarke G, Shanahan F, Dinan TG, Cryan JF, O’Leary OF. Adult hippocampal neurogenesis is regulated by the microbiome. Biol Psychiatry. 2015 Aug 15;78(4):e7–9.
- Heiss CN, Olofsson LE. Gut Microbiota-Dependent Modulation of Energy Metabolism. J Innate Immun. 2018;10(3):163-171.
- Koh A, De Vadder F, Kovatcheva-Datchary P, Bäckhed F. From Dietary Fiber to Host Physiology: Short-Chain Fatty Acids as Key Bacterial Metabolites. Cell. 2016 Jun 2;165(6):1332–1345.
- Holzer P, Reichmann F, Farzi A. Neuropeptide Y, peptide YY and pancreatic polypeptide in the gut-brain axis. Neuropeptides. 2012 Dec;46(6):261–274.
- Mawe GM, Hoffman JM. Serotonin signaling in the gut–functions, dysfunctions and therapeutic targets. Nat Rev Gastroenterol Hepatol. 2013 Aug;10(8):473–86.
- Thijssen AY, Mujagic Z, Jonkers DMAE, Ludidi S, Keszthelyi D, Hesselink MA, et al. Alterations in serotonin metabolism in the irritable bowel syndrome. Aliment Pharmacol Ther. 2016 Jan;43(2):272–82.
- Kau AL, Ahern PP, Griffin NW, Goodman AL, Gordon JI. Human nutrition, the gut microbiome and the immune system [Internet]. Vol. 474, Nature. p. 327–36. Available from: http://dx.doi.org/10.1038/nature10213
- Leclercq S, Matamoros S, Cani PD, Neyrinck AM, Jamar F, Starkel P, et al. Intestinal permeability, gut-bacterial dysbiosis, and behavioral markers of alcohol-dependence severity. Proc Natl Acad Sci USA. 2014 Oct 21;111(42)E4485–E4493.
- Jansen JM, van Holst RJ, van den Brink W, Veltman DJ, Caan MWA, Goudriaan AE. Brain function during cognitive flexibility and white matter integrity in alcohol-dependent patients, problematic drinkers and healthy controls. Addict Biol. 2015 Sep;20(5):979–989.
- Fan Y, Ya-E Z, Ji-Dong W, Yu-Fan L, Ying Z, Ya-Lun S, et al. Comparison of microbial diversity and composition in jejunum and colon of the alcohol-dependent rats. J Microbiol Biotechnol. 2018 Nov 28;28(11):1883–1895.
- Bajaj JS. Alcohol, liver disease and the gut microbiota [Internet]. Vol. 16, Nature Reviews Gastroenterology & Hepatology. 2019. p. 235–246. Available from: http://dx.doi.org/10.1038/s41575- 018-0099-1
- Cani PD, Possemiers S, Van de Wiele T, Guiot Y, Everard A, Rottier O, et al. Changes in gut microbiota control inflammation in obese mice through a mechanism involving GLP-2- driven improvement of gut permeability. Gut. 2009 Aug;58(8):1091–1103.
- Chen PB, Yang JS, Park Y. Adaptations of Skeletal Muscle Mitochondria to Obesity, Exercise, and Polyunsaturated Fatty Acids. Lipids. 2018 Mar;53(3):271-278.
- Drake JC, Wilson RJ, Yan Z. Molecular mechanisms for mitochondrial adaptation to exercise training in skeletal muscle [Internet]. Vol. 30, The FASEB Journal. 2016. p.13-22. Available from: http://dx.doi.org/10.1096/fj.15-276337
- Petersen AMW, Pedersen BK. The anti-inflammatory effect of exercise [Internet]. Vol. 98, Journal of Applied Physiology. 2005. p. 1154-1162. Available from: http://dx.doi.org/10.1152/japplphysiol.00164.2004
- Correia SS, Goosens KA. Input-specific contributions to valence processing in the amygdala. Learn Mem. 2016 Oct;23(10):534–543.
- Cai YQ, Wang W, Paulucci-Holthauzen A, Pan ZZ. Brain circuits mediating opposing effects on emotion and pain. J Neurosci. 2018 Jul 11;38(28):6340–6349.
- Sherwin E, Bordenstein SR, Quinn JL, Dinan TG, Cryan JF. Microbiota and the social brain. Science [Internet]. 2019 Nov 1;366(6465). Available from: http://dx.doi.org/10.1126/science.aar2016
- Qin HY. Impact of psychological stress on irritable bowel syndrome [Internet]. Vol. 20, World Journal of Gastroenterology. 2014. p. 14126–14131. Available from: http://dx.doi.org/10.3748/wjg.v20.i39.14126
- Rohleder N, Aringer M, Boentert M. Role of interleukin-6 in stress, sleep, and fatigue. Ann N Y Acad Sci. 2012 Jul;1261:88-96.
- Aguilera M, Vergara P, Martínez V. Stress and antibiotics alter luminal and wall adhered microbiota and enhance the local expression of visceral sensory-related systems in mice. Neurogastroenterol Motil. 2013 Aug;25(8):e515–e529.
- Konturek PC, Brzozowski T, Konturek SJ. Stress and the gut: pathophysiology, clinical consequences, diagnostic approach and treatment options. J Physiol Pharmacol. 2011 Dec;62(6):591– 599.
- Karl JP, Margolis LM, Madslien EH, Murphy NE, Castellani JW, Gundersen Y, et al. Changes in intestinal microbiota composition and metabolism coincide with increased intestinal permeability in young adults under prolonged physiological stress. Am J Physiol Gastrointest Liver Physiol. 2017 Jun 1;312(6):G559-571.
- Blanchard EB, Lackner JM, Jaccard J, Rowell D, Carosella AM, Powell C, et al. The role of stress in symptom exacerbation among IBS patients. J Psychosom Res. 2008 Feb;64(2):119-128.
- Chang L. The role of stress on physiologic responses and clinical symptoms in irritable bowel syndrome. Gastroenterology. 2011 Mar;140(3):761–765.
- Stasi C, Rosselli M, Bellini M, Laffi G, Milani S. Altered neuro-endocrine-immune pathways in the irritable bowel syndrome: the top-down and the bottom-up model. J Gastroenterol. 2012 Nov;47(11):1177–1185.
- Sripada CS, Angstadt M, McNamara P, King AC, Phan KL. Effects of alcohol on brain responses to social signals of threat in humans. Neuroimage. 2011 Mar 1;55(1):371–380.
- Aloi J, Blair KS, Crum KI, Meffert H, White SF, Tyler PM, et al. Adolescents show differential dysfunctions related to Alcohol and Cannabis Use Disorder severity in emotion and executive attention neuro-circuitries. Neuroimage Clin. 2018 Jun 5;19:782–792.
- Kelly JR, Kennedy PJ, Cryan JF, Dinan TG, Clarke G, Hyland NP. Breaking down the barriers: the gut microbiome, intestinal permeability and stress-related psychiatric disorders. Front Cell Neurosci. 2015 Oct 14;9:392.
- Elsenbruch S. Abdominal pain in Irritable Bowel Syndrome: A review of putative psychological, neural and neuro-immune mechanisms. Brain Behav Immun. 2011 Mar;25(3):386-394.
- Bailey MT, Dowd SE, Galley JD, Hufnagle AR, Allen RG, Lyte M. Exposure to a social stressor alters the structure of the intestinal microbiota: implications for stressor induced immunomodulation. Brain Behav Immun. 2011 Mar;25(3):397–407.
- Noguera JC, Aira M, Perez-Losada M, Dominguez J,Velando A.Glucocorticoids modulate gastrointestinal microbiome in a wild bird.Royal Society Open Science.2018;1-8
- Bharwani A, Mian MF, Foster JA, Surette MG, Bienenstock J, Forsythe P. Structural & functional consequences of chronic psychosocial stress on the microbiome & host. Psychoneuroendocrinology. 2016 Jan;63:217‐227.
- Foster JA, Rinaman L, Cryan JF. Stress & the gut‐brain axis: Regulation by the microbiome [Internet]. Vol. 7, Neurobiology of Stress. 2017. p. 124‐136. Available from:
http://dx.doi.org/10.1016/j.ynstr.2017.03.001 - O’Mahony SM, Marchesi JR, Scully P, Codling C, Ceolho AM, Quigley EMM, et al. Early life stress alters behavior, immunity, and microbiota in rats: Implications for irritable bowel syndrome and psychiatric illnesses. Biol Psychiatry. 2009 Feb 1;65(3):263–7.
- Sudo N. Stress and gut microbiota: Does postnatal microbial colonization program the hypothalamic-pituitary-adrenal system for stress response? [Internet]. Vo. 1287, International Congress Series. 2006. p.350–4. Available from: http://dx.doi.org/10.1016/j.ics.2005.12.019
- Desbonnet L, Garrett L, Clarke G, Bienenstock J, Dinan TG. The probiotic Bifidobacteria infantis: An assessment of potential antidepressant properties in the rat. J Psychiatr Res. 2008 Dec;43(2):164– 74.
- Barouei J, Moussavi M, Hodgson DM. Effect of maternal probiotic intervention on HPA axis, immunity and gut microbiota in a rat model of irritable bowel syndrome [Internet]. Vol. 7, PLoS One. 2012. p. e46051. Available from: http://dx.doi.org/10.1371/journal.pone.0046051
- McCusker RH, Kelley KW. Immune-neural connections: How the immune system’s response to infectious agents influences behavior [Internet]. Vol. 216, Journal of Experimental Biology. 2013. p. 84–98. Available from: http://dx.doi.org/10.1242/jeb.073411
- Moreira CG, Russell R, Mishra AA, Narayanan S, Ritchie JM, Waldor MK, et al. Bacterial adrenergic sensors regulate virulence of enteric pathogens in the gut. MBio [Internet]. 2016 Jun 7;7(3). Available from: http://dx.doi.org/10.1128/mBio.00826-16
- Zamani M, Alizadeh-Tabari S, Zamani V. Systematic review with meta-analysis: the prevalence of anxiety and depression in patients with irritable bowel syndrome. Aliment Pharmacol Ther. 2019 Jul;50(2):132–143.
- Halland M, Almazar A, Lee R, Atkinson E, Larson J, Talley NJ, et al. A case-control study of childhood trauma in the development of irritable bowel syndrome. Neurogastroenterol Motil. 2014 Jul;26(7):990–8.
- Sykes MA, Blanchard EB, Lackner J, Keefer L, Krasner S. Psychopathology in irritable bowel syndrome: support for a psychophysiological model. J Behav Med. 2003 Aug;26(4):361–72.
- Alonso C, Guilarte M, Vicario M, Ramos L, Ramadan Z, Antolín M, et al. Maladaptive intestinal epithelial responses to life stress may predispose healthy women to gut mucosal inflammation. Gastroenterology. 2008 Jul;135(1):163-172.e1.
- Söderholm JD, Yang PC, Ceponis P, Vohra A, Riddell R, Sherman PM, et al. Chronic stress induces mast cell-dependent bacterial adherence and initiates mucosal inflammation in rat intestine.
Gastroenterology. 2002 Oct;123(4):1099-1108. - Barbara G, WangB, Stanghellini V, de Giorgio R, Cremon C, Di Nardo G, et al. Mast cell- dependent excitation of visceral-nociceptive sensory neurons in irritable bowel syndrome. Gastroenterology. 2007 Jan;132(1):26-37.
- Cenac N, Andrews CN, Holzhausen M, Chapman K, Cottrell G, Andrade-Gordon P, et al. Role for protease activity in visceral pain in irritable bowel syndrome. J Clin Invest. 2007 Mar;117(3): 636- 647.
- Santos J, Benjamin M, Yang PC, Prior T, Perdue MH. Chronic stress impairs rat growth and jejunal epithelial barrier function: role of mast cells. Am J Physiol Gastrointest Liver Physiol. 2000 Jun;278(6):G847-G854.
- Meddings JB, Swain MG. Environmental stress-induced gastrointestinal permeability is mediated by endogenous glucocorticoids in the rat. Gastroenterology. 2000 Oct; 119(4):1019– 1028.
- Söderholm JD, Perdue MH. Stress and gastrointestinal tract. II. Stress and intestinal barrier function [Internet]. Vol. 280, American Journal of Physiology-Gastrointestinal and Liver Physiology. 2001. p. G7-G13. Available from: http://dx.doi.org/10.1152/ajpgi.2001.280.1.g7
- Vanuytsel T, van Wanrooy S, Vanhell H, Vanormelingen C, Verschueren S, Houben E, et al. Psychological stress and corticotropin-releasing hormone increase intestinal permeability in humans by a mast cell-dependent mechanism. Gut. 2014 Aug;63(8):1293- 1299.
- Söderholm JD, Yang PC, Ceponis P, Vohra A, Riddell R, Sherman PM, Perdue MH . Chronic stress induces mast cell-dependent bacterial adherence and initiates mucosal inflammation in rat intestine. Gastroenterology. 2002 Oct;123(4):1099-1108. doi: 10.1053/gast.2002.36019.
- Santos J, Saperas E, Nogueiras C, Mourelle M, Antolín M, Cadahia A, et al. Release of mast cell mediators into the jejunum by cold pain stress in humans. Gastroenterology. 1998 Apr;114(4):640- 648.
- Eutamene H, Theodorou V, Fioramonti J, Bueno L. Acute stress modulates the histamine content of mast cells in the gastrointestinal tract through interleukin-1 and corticotropin-releasing factor release in rats. J Physiol. 2003 Dec 15;553(Pt 3):959-966.
- Santos J, Yang PC, Söderholm JD, Benjamin M, Perdue MH. Role of mast cells in chronic stress induced colonic epithelial barrier dysfunction in the rat. Gut. 2001 May;48(5):630-636
- Lyte M, Ernst S. Catecholamine induced growth of gram negative bacteria [Internet]. Vol. 50, Life Sciences. 1992. p. 203-212. Available from: http://dx.doi.org/10.1016/0024- 3205(92)90273-r
- Barclay GR, Turnberg LA. Effect of psychological stress on salt and water transport in the human jejunum. Gastroenterology. 1987 Jul;93(1):91-97.
- Stout C, Snyder RL. Ulcerative colitis-like lesion in Siamang gibbons. Gastroenterology. 1969 Sep;57(3):256-261.
- Ye L, Liddle RA. Gastrointestinal hormones and the gut connectome. Curr Opin Endocrinol Diabetes Obes. 2017 Feb;24(1):9–14.
- André P, Laugerette F, Féart C. Metabolic Endotoxemia: A Potential Underlying Mechanism of the Relationship between Dietary Fat Intake and Risk for Cognitive Impairments in Humans? Nutrients [Internet]. 2019 Aug 13;11(8). Available from: http://dx.doi.org/10.3390/nu11081887
- Chang L, Toner BB, Fukudo S, Guthrie E, Richard Locke G, Norton NJ, et al. Gender, age, society, culture, and the patient’s perspective in the functional gastrointestinal disorders [Internet]. Vol. 130, Gastroenterology. 2006. p.1435-1446. Available from: http://dx.doi.org/10.1053/j.gastro.2005.09.071
- Moreira AP, Texeira TF, Ferreira AB, Peluzio M do CG, Alfenas R de CG. Influence of a high-fat diet on gut microbiota, intestinal permeability and metabolic endotoxaemia. Br J Nutr. 2012 Sep;108(5):801–809.
- Fei N, Zhao L. An opportunistic pathogen isolated from the gut of an obese human causes obesity in germfree mice. ISME J. 2013 Apr;7(4):880–84.
- Kim KA, Gu W, Lee IA, Joh EH, Kim DH. High fat diet-induced gut microbiota exacerbates inflammation and obesity in mice via the TLR4 signaling pathway. PLoS One. 2012 Oct 16;7(10):e47713.
- Costabile A, Klinder A, Fava F, Napolitano A, Fogliano V, Leonard C, et al. Whole-grain wheat breakfast cereal has a prebiotic effect on the human gut microbiota: a double-blind, placebo-controlled, crossover study [Internet]. Vol. 99, British Journal of Nutrition. 2008. p. 110–20. Available from: http://dx.doi.org/10.1017/s0007114507793923
- De Filippo C, Cavalieri D, Di Paola M, Ramazzotti M, Poullet JB, Massart S, et al. Impact of diet in shaping gut microbiota revealed by a comparative study in children from Europe and rural Africa. Proc Natl Acad Sci USA. 2010 Aug 17;107(33):14691–6.
- Cryan JF, Dinan TG. Mind-altering microorganisms: the impact of the gut microbiota on brain and behaviour. Nat Rev Neurosci. 2012 Oct;13(10):701–12.
- Lozupone CA, Stombaugh JI, Gordon JI, Jansson JK, Knight R. Diversity, stability and resilience of the human gut microbiota. Nature. 2012 Sep 13;489(7415):220‐230.
- Obata Y, Pachnis V. The Effect of Microbiota and the Immune System on the Development and Organization of the Enteric Nervous System. Gastroenterology 2016 Nov;151(5):836–844.
- Reigstad CS, Salmonson CE, Rainey JF 3rd, Szurszewski JH, Linden DR, Sonnenburg JL, et al. Gut microbes promote colonic serotonin production through an effect of short-chain fatty acids on enterochromaffin cells. FASEB J. 2015 Apr;29(4):1395–403.
- Mayer EA. Gut feelings: the emerging biology of gut‐brain communication. Nat Rev Neurosci. 2011 Jul 13;12(8):453‐466.
- Luczynski P, Neufeld KA, Oriach CS, Clarke G, Dinan TG, Cryan JF. Growing up in a Bubble: Using Germ-Free Animals to Assess the Influence of the Gut Microbiota on Brain and Behavior [Internet]. Vol. 19, International Journal of Neuropsychopharmacology. 2016. p. yw020. Available from: http://dx.doi.org/10.1093/ijnp/pyw020
- Khoshdel A, Verdu EF, Kunze W, McLean P, Bergonzelli G, Huizinga JD. Bifidobacterium longum NCC3001 inhibits AH neuron excitability. Neurogastroenterol Motil. 2013 Jul;25(7):e478– 84.
- Blakemore, SJ. (2008). The social brain in adolescence. Nat. Rev. Neurosci. 2008 ;9(4) : 267– 277. doi: 10.1038/nrn2353.
- Hillemacher, T, Bachmann O, Kahl KG, Frieling H. Alcohol, microbiome, and their effect on psychiatric disorders [Internet]. Vol. 85, Progress Neuro-Psychopharmacology and Biologyical Psychiatry. 2008. p. 105–115. Available from: http://dx.doi.org/10.1016/j.pnpbp.2018.04.015
- Yu CD, Xu QJ, Chang RB. Vagal sensory neurons and gut-brain signaling. Curr Opin Neurobiol. 2020 Jun;62:133–140.
- Camilleri, M. Serotonin in the gastrointestinal tract [Internet]. Vol. 16, Current Opinion in Endocrinology, Diabetes and Obesity. 2009. p. 53–59. Available from: http://dx.doi.org/10.1097/med.0b013e32831e9c8e
- Gronier B, Savignac HM, Di Miceli M, Idriss SM, Tzortzis G, Anthony D, et al. Increased cortical neuronal responses to NMDA and improved attentional set-shifting performance in rats following prebiotic (B-GOS) ingestion. Eur Neuropsychopharmacol. 2018 Jan;28 (1):211–224.
- Strandwitz P, Kim KH, Terekhova D, Liu JK, Sharma A, Levering J, et al. GABA modulating bacteria of the human gut microbiota. Nat. Microbiol. 2019 Mar;4(3):396–403.
- Khachatryan ZA, Ktsoyan ZA, Manukyan GP, Kelly D, Ghazaryan KA, Aminov RI. Predominant role of host genetics in controlling the composition of gut microbiota. PLoS One. 2008 Aug 26;3(8):e3064.
- De Filippo C, Cavalieri D, Di Paola M, Ramazzotti M, Poullet JB, Massart S, et al. Impact of diet in shaping gut microbiota revealed by a comparative study in children from Europe and rural Africa. Proc Natl Acad Sci USA. 2010 Aug 17;107(33):14691–6.
- Nam YD, Jung MJ, Roh SW, Kim MS, Bae JW. Comparative analysis of Korean human gut microbiota by barcoded pyrosequencing. PLoS One. 2011 Jul 29;6(7):e22109.
- Kim JS, De la Serre CB. Diet, gut microbiota composition and feeding behavior.Physiology & Behavior.2018;192:177-181
- Long-Smith C, O’Riordan KJ, Clarke G, Stanton C, Dinan TG, Cryan JF. Microbiota Gut-Brain Axis: New Therapeutic Opportunities. Annu Rev Pharmacol Toxicol. 2020 Jan 6;60:477–502.
- Zheng P, Zeng B, Zhou C, Liu M, Fang Z, Xu X, et al. Gut microbiome remodeling induces depressive-like behaviors through a pathway mediated by the host’s metabolism [Internet]. Vol. 21, Molecular Psychiatry. 2016. p. 786–96. Available from: http://dx.doi.org/10.1038/mp.2016.44
- Russell SL, Gold MJ, Hartmann M, Willing BP, Thorson L, Wlodarska M, et al. Early life antibiotic‐driven changes in microbiota enhance susceptibility to allergic asthma [Internet]. Vol. 13, EMBO Reports. 2012. p. 440‐447. Available from: http://dx.doi.org/10.1038/embor.2012.32
- Gonzalez A, Stombaugh J, Lozupone C, Turnbaugh PJ, Gordon JI, Knight R. The mind‐body‐ microbial continuum. Dialogues Clin Neurosci. 2011;13(1):55‐62.
- Sharma VR,Singh M, Kurman V, Yadav M, Nirmala S, Sharma D, Sharma A. . Microbiome dysbiosis in cancer:Exploring therapeutic strategies to counter the disease. Seminars in Cancer Biology.2021;70:61-70
- Haslam DW, James WPT. Obesity. Lancet. 2005 Oct 1;366(9492):1197‐1209.
- Sandhu KV, Sherwin E, Schellekens H, Stanton C, Dinan TG, Cryan JF. Feeding the microbiota‐gut‐brain axis: diet, microbiome, and neuropsychiatry. Transl Res. 2017 Jan;179:223‐244.
- Krajmalnik‐Brown R, Ilhan ZE, Kang DW, DiBaise JK. Effects of gut microbes on nutrient absorption and energy regulation. Nutr Clin Pract. 2012 Apr;27(2):201‐214.
- Labanski A, Langhorst J, Engler H, Elsenbruch S. Stress and the brain-gut axis in functional and chronic-inflammatory gastrointestinal diseases: a transdisciplinary challenge. Psychoneuroendocrinology. 2020 Jan;111:104501.
- Enck P, Aziz Q, Barbara G, Farmer AD, Fukudo S, Mayer EA, et al. Irritable bowel syndrome. Nat Rev Dis Primers. 2016 Mar 24;2:16014.
- Chassaing B, Koren O, Goodrich JK, Poole AC, Srinivasan S, Ley RE, et al. Dietary emulsifiers impact the mouse gut microbiota promoting colitis and metabolic syndrome. Nature. 2015 Mar 5;519(7541):92‐96.
- Beck, AT. Cognitive Therapy and the emotional disorders. International Universities PressInc; 1976. 356 p.
- Barnes-Holmes Y, Hayes SC, Barnes-Holmes D, Roche, B. Relational frame theory: A post- skinnerian account of human language and cognition [Internet]. Advances in Child Development and Behaviour. 2002. p. 101-38. Available from: http://dx.doi.org/10.1016/s0065- 2407(02)80063
- Kabat-Zinn J. An outpatient program in behavioral medicine for chronic pain patients based on the practice of mindfulness meditation: Theoretical consideration and preliminary results. Gen Hosp Psychiatry.1982 Apr;4(1):33- 47.
- Kegan R, Lahey LL. Immunity to Change: How to Overcome It and Unlock the Potential in Yourself and Your Organization. Harvard Business Review Press; 2009. 359 p.
- Prochaska JO, DiClemente CC. The transtheoretical approach [Internet]. Handbook of Psychotherapy Integration. 2005. p. 147-71. Available from: http://dx.doi.org/10.1093/med:psych/9780195165791.003.0007
- Nistico V, Rossi R, D Arrigo A,Priori A, Gambini O, Demartini B.Functional Neuroimaging in Irritable Bowel Syndrome: A Systematic Review Highlights Common Brain Alterations with Fuctional Movement Disorders.J Neurogastroenterol Motil.2022;28(2):185- 203
- Smith R, Badcock P, Friston K.Recent advances in the applicationof predictive coding and active inference models within clinical neuroscience.Psychiatriy and Clinical Neurosciences.2020;75(1:3-13
- Ainley V, Apps M, Fotopoulou A, Tsakiris M.Bodily precision:a predictive coding account of individual differences in interoceptive accuracy.Philos Trans Soc Lond B Biol Sci.2016;19:371(1708)20160003
- Felman L, Quigley K,Hamilton P. An active inference theory of allostasis and inroception in depression. Philos Trans Soc Lond B Biol.2016;371:20160011
- Ohira H. Regulaion of functions of Brain and body by the principle of predictive coding:implications for impairments of the brain-gut axis.Psychological Topics.2018;27:1-15
- Peter Senge. The fifth discipline. Doubleday; Revised and Updated edition (2006)
- Seligman MEP, Csikszentmihalyi M. Positive psychology: An introduction. American Psychologist, 2000 ;55(1): 5–14.doi :10.1037//0003-066x.55.1.5.
- Frankl VE. Man’s search for meaning. Gift Edition. Beacon Press; 2004. 202 p.
- Cani PD, Jordan BF. Gut microbiota-mediated inflammation in obesity: a link with gastrointestinal cancer. Nat Rev Gasroenterol Hepatol. 2018 Nov;15(11):671-682.
- Kovatcheva-Datchary P,Arora T, Nutrition , the gut microbiome and the metabolic syndrome. Best Pract res Clin Gastroenterol.2013;27(1):59-72.doi: 10.1016/j.bpg.2013.03.017
- Gallard L. The gut microbiome and the brain. Journal Medical Food. 2014;17(12):1261-1272.
- Lombardi VC, De Meirleir KL, Subramanian K, Nourani SM, Dagda RK, Delaney SL, et al. Nutritional Modulation of the Intestinal Microbiota; Future Opportunitiesfor the Prevention andTreatment of Neuroimmune and Neuroinflammatory Disease. J Nutr Biochem. 2018 Nov;61:1–16.
- Caesar R, Fåk F, Bäckhed F. Effects of gut microbiota on obesity and atherosclerosis via modulation of inflammation and lipid metabolism [Internet]. Vol. 268, Journal of Internal Medicine. 2010. p. 320-328. Available from: http://dx.doi.org/10.1111/j.1365- 2796.2010.02270.x
- Lima-Ojeda JM, Rupprecht R, Baghai C. “I Am I and My Bacterial Circumstances”: Linking Gut Microbiome, Neurodevelopment, and Depression. Front Psychiatry. 2017 Aug 22;8:153.
- Turnbaugh PJ, Hamady M, Yatsunenko T, Cantarel BL, Duncan A, Ley RE, et al. A core gut microbiome in obese and lean twins. Nature. 2009 Jan 22;457(7228):480–4.
- Bindels LB, Delzenne NM, Cani PD, Walter J. Towards a more comprehensive concept for prebiotics. Nat Rev Gastroenterol Hepatol. 2015 May;12(5):303‐310.
- Torres‐Fuentes C, Schellekens H, Dinan TG, Cryan JF. A natural solution for obesity: Bioactives for the prevention and treatment of weight gain. A Review. Nutr Neurosci. 2015 Feb;18(2):49‐65.
- Cani PD, Osto M, Geurts L, Everard A. Involvement of gut microbiota in the developmental low-grade inflammation and type 2 diabetes associated with obesity [Internet]. Vol. 3, Gut microbes. 2012. p. 279-288. Available from: http://dx.doi.org/10.4161/gmic.19625
- Spinler JK, Taweechotipatr M, Rognerud CL, Ou CN, Tumwasorn S, Versalovic J. Human- derived probiotic Lactobacillus reuteri demonstrate antimicrobial activities targeting diverse enteric bacterial pathogens. Anaerobe. 2008 Jun;14(3):166–
- Álvarez‐Arraño V, Martín‐Peláez S. Effects of probiotics and synbiotics on weight loss in subjects with overweight or obesity: A systematic review [Internet]. Nutrients. 2021. p. 3627. Available from: http://dx.doi.org/10.3390/nu13103627
- Asadi A, Mehr NS, Mohamadi MH, Shokri F, Heidary M, Sadeghifard N, et al. Obesity and gut- microbiota-brain axis: A narrative review [Internet]. Vol. 36, Journal of Clinical Laboratory Analysis. 2022. Available from: http://dx.doi.org/10.1002/jcla.24420
- Thomas CM, Versalovic J. Probiotics-host communication: modulation of signaling pathways in the intestine. Gut Microbes. 2010 May;1(3):148–63.
- Guarner F, Khan AG, Garisch J, Eliakim R, Gangl A, Thomson A, et al. World gastroenterology organisation global guidelines: probiotics and prebiotics October 2011. J Clin Gastroenterol. 2012 Jul;46(6):468‐481.
- Sommer F, Bäckhed F. The gut microbiota—masters of host development and physiology. Nat Rev Microbiol. 2013 Apr;11(4):227–38.
- de Vrese M, Schrezenmeir J. Probiotics, prebiotics, and synbiotics. Adv Biochem Eng Biotechnol.2008;111:1-66.doi:10.1007/10_2008_097.
- Pandey KR, Naik SR, Vakil BV. Probiotics, prebiotics and synbiotics‐ a review. J Food Sci Technol. 2015 Dec;52(12):7577‐7587.
- Khoruts A, Sadowsky MJ. Understanding the mechanisms of faecal microbiota transplantation [Internet]. Vol. 13, Nature Reviews Gastroenterology & Hepatology. 2016. p. 508‐516. Available from: http://dx.doi.org/10.1038/nrgastro.2016.98
- Chen PB, Yang JS, Park Y. Adaptations of skeletal muscle mitochondria to obesity, exercise, and polyunsaturated fatty acids. Lipids. 2018;53:271-278.


 Figure 3: Imbalance of GBNM2 System
Figure 3: Imbalance of GBNM2 System